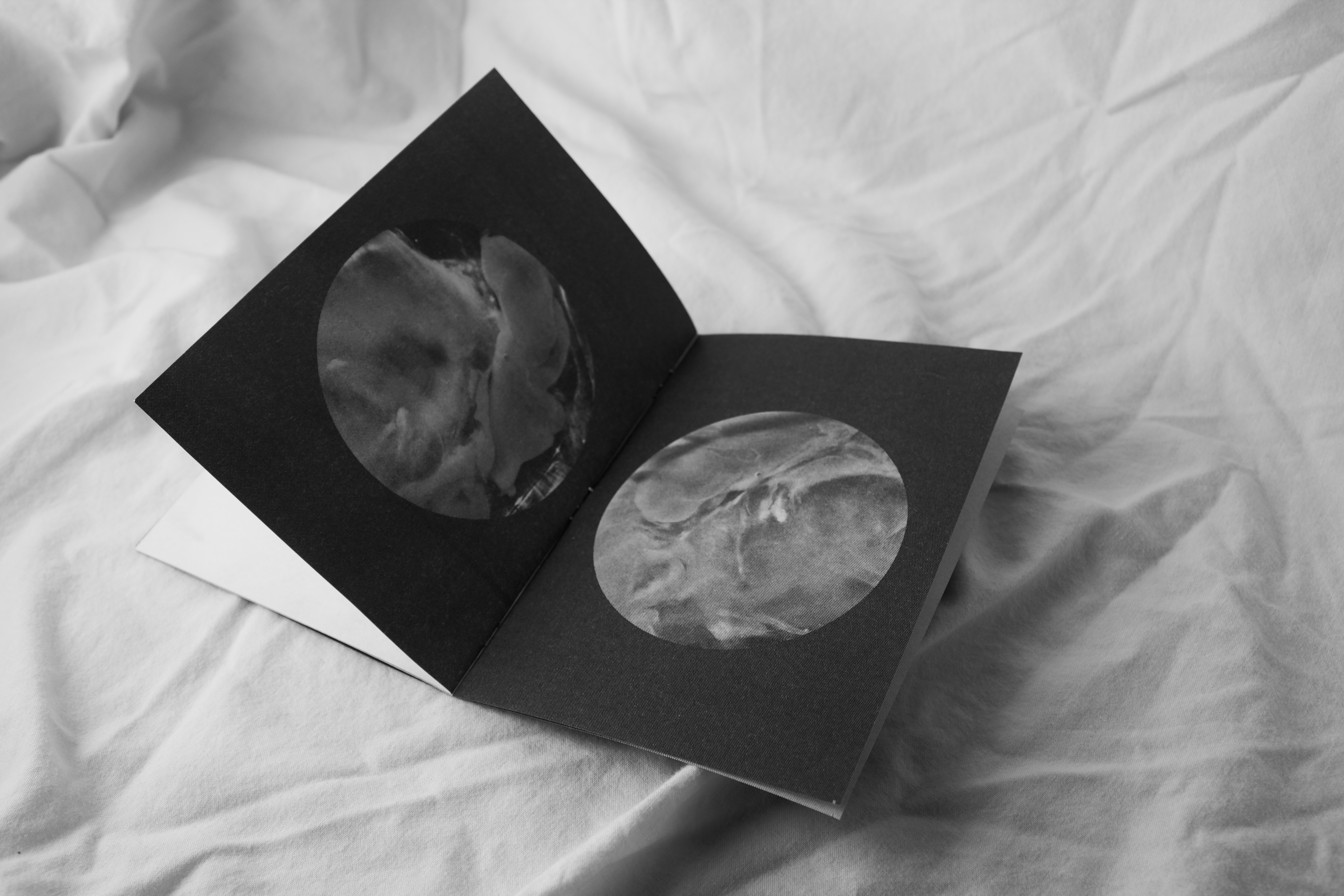
The second law of thermodynamics, entropy, is commonly simplified through the analogy of an egg.
An egg in its original and whole form is an ordered system and has a low level of entropy. But once this egg is cracked an irreversible process has taken place and the entropy of the egg has increased.
The object has become disordered and can never revert back to its ordered state. Thus illustrating the idea that entropy is ever increasing through the passage of time. A Publication designed to go alongside the exhibition and explain to the viewer the concept behind the exhibition itself.
The goal of this exhibition space is to allow the viewer to be immersed in an entropic environment in which the can follow these “irreversible cracks” and understand the idea of entropy through the physical disorder within the space
A Publication designed to go alongside the exhibition and explain to the viewer the concept behind the exhibition itself.
The goal of this exhibition space is to allow the viewer to be immersed in an entropic environment in which the can follow these “irreversible cracks” and understand the idea of entropy through the physical disorder within the space.